BIOELECTRONICS & DIAGNOSTICS .
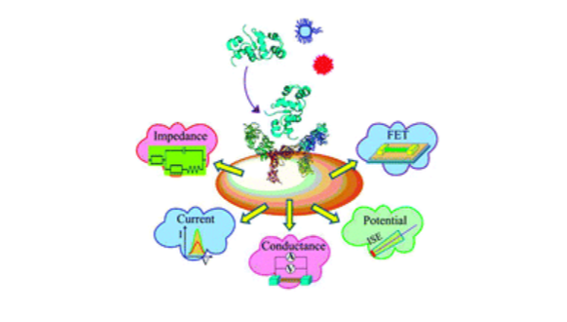
The group actively develop and apply new electroanalytical methods capable of tracking the molecular recognition that underpins diagnostics
These have included new capacitative, shotgun, enzymic and kinetic methods of sampling molecular targets. Some of these are exquisitely (fM) sensitive and, through integration within designed and 3D printed microfluidic configurations, generate readouts within minutes.
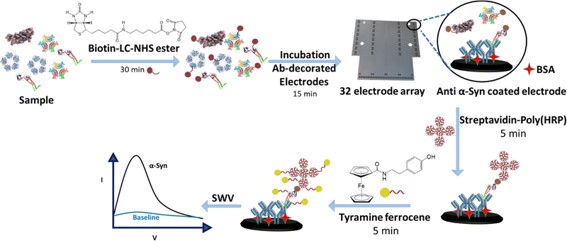
We also develop new capture nanoparticle chemistries and have applied some of these with magnetically assisted microfluidics to further optimise target capture from patient blood. We work with teams at the Oxford University NHS Trust, Molecular Oncology, Neurology, the Nuffield Department of Orthopaedics and the Nuffield Department of Surgical Sciences.




EXAMPLES .
Ultrafast Biomarker Quantification through Reagentless Capacitive Kinetics
Anal. Chem. 2023, 95, 4721-4727–7913
Online Paper
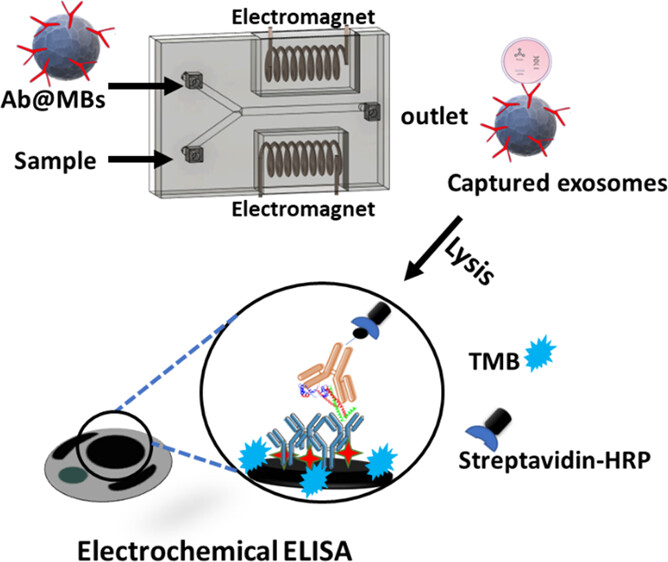
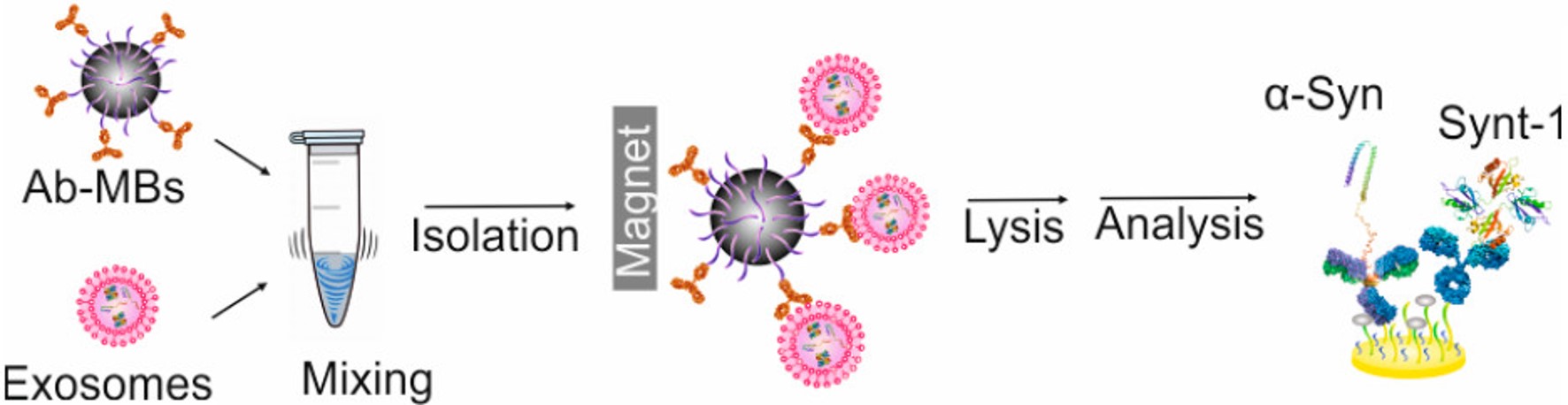
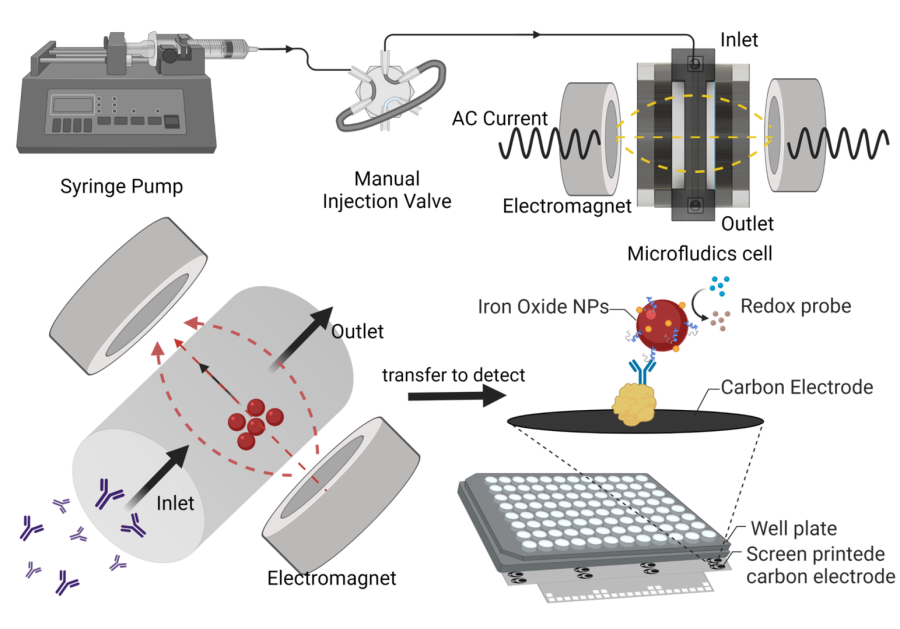
Alternating Magnetic Field-Promoted Nanoparticle Mixing: The On-Chip Immunocapture of Serum Neuronal Exosomes for Parkinson's Disease Diagnostics
Anal. Chem. 2023, 96, 7906-7913
Online Paper
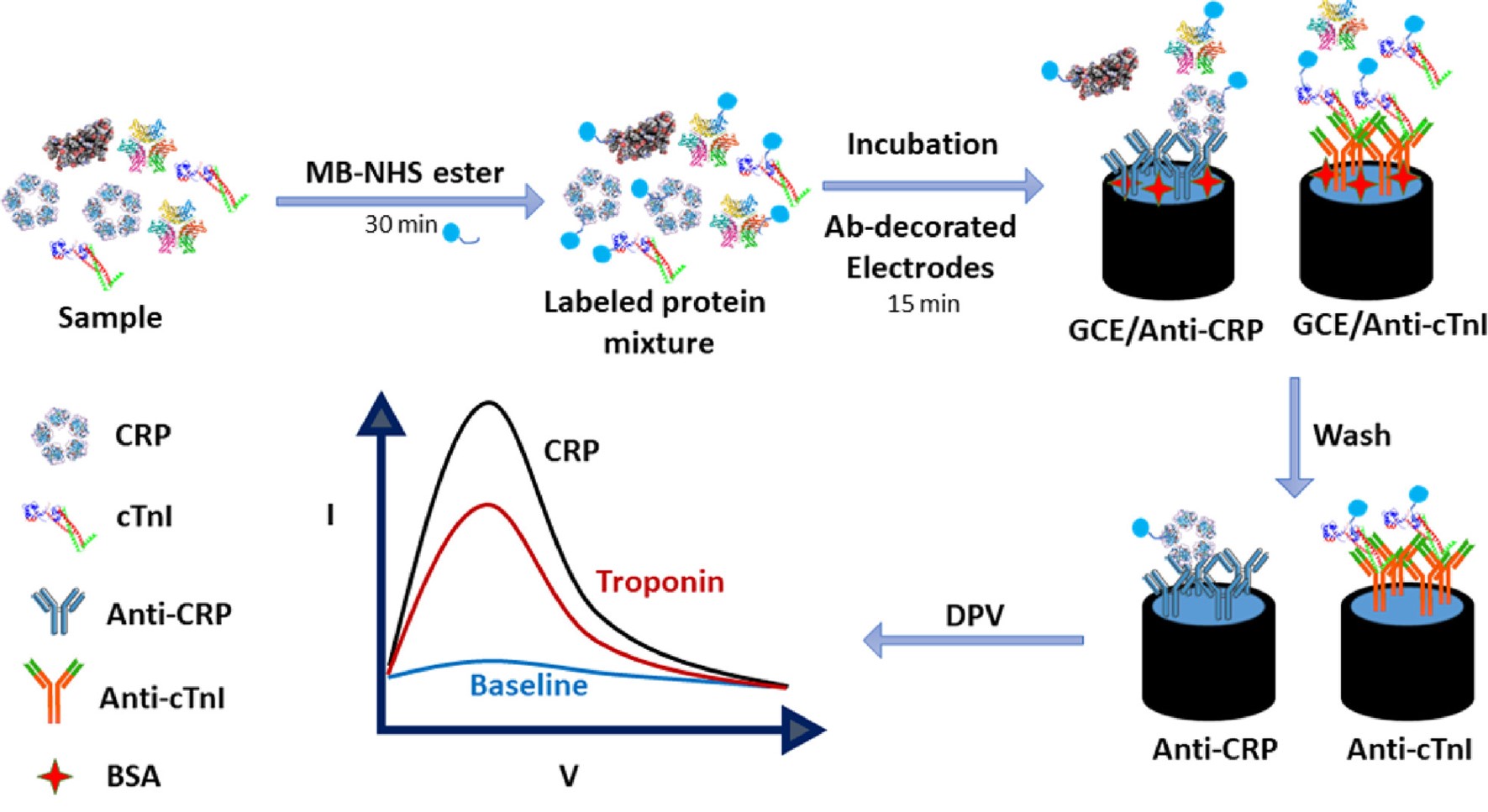
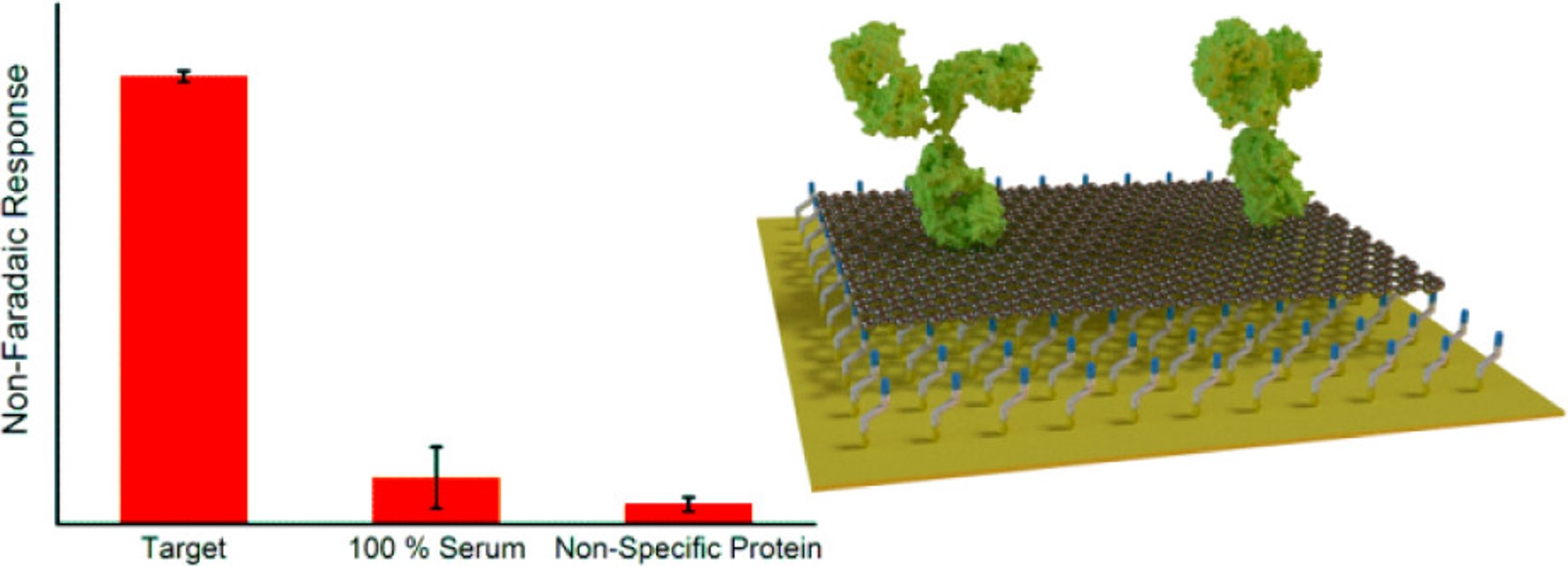
Catalysed amplification of faradaic shotgun tagging in ultrasensitive electrochemical immunoassays
Chem. Commun., 2022, 58, 9472-9475;
Online Paper
A quantification of target protein biomarkers in complex media in faradaic shotgun tagging
Anal. Chem., 2022, 94, 2375-2382;
Online Paper
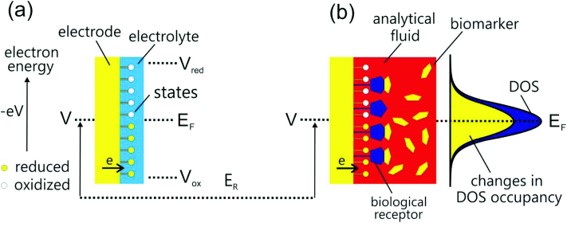
Quantum capacitance as a reagentless molecular sensing element
Nanoscale, 2017, 9, 15362-15370;
Online Paper
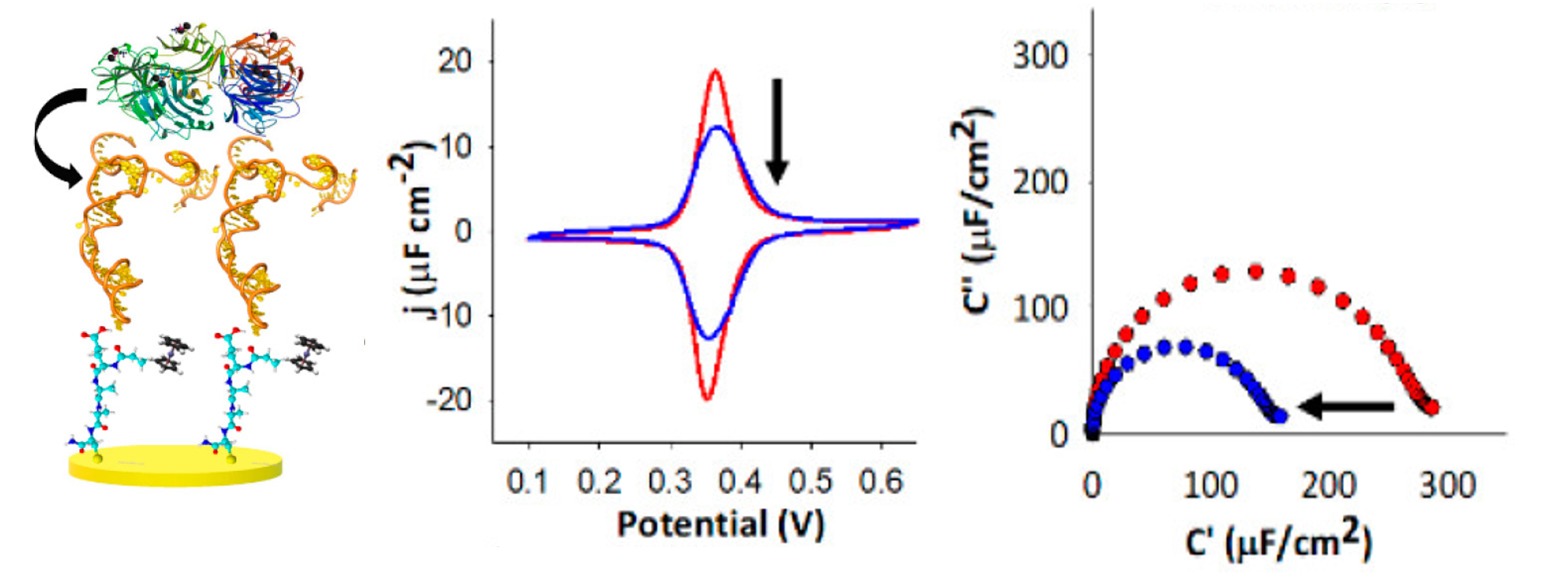
Redox Capacitive Assaying of C-Reactive Protein at a Peptide Supported Aptamer Interface
Anal. Chem., 2018, 90, 3005-3008;
Online Paper
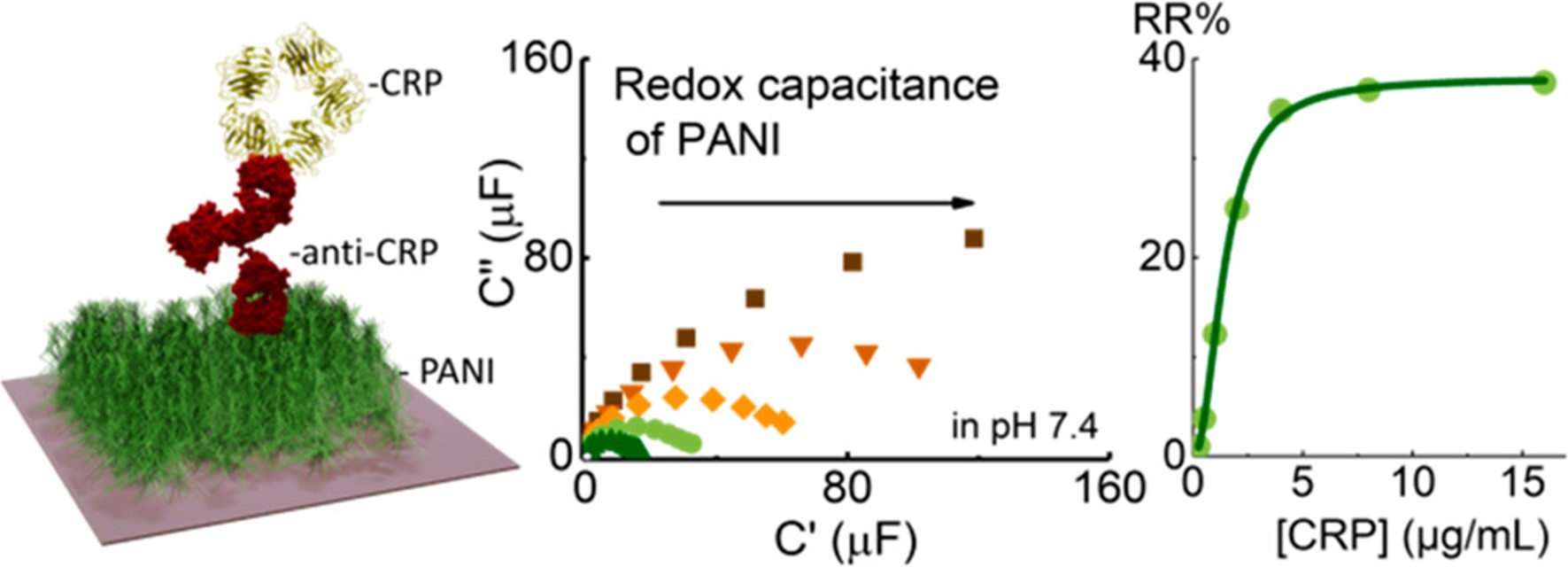
Reagentless Redox Capacitive Assaying of C-Reactive Protein at a Polyaniline Interface
Anal. Chem., 2020, 92, 3508-3511;
Online Paper
Facile Impedimetric Analysis of Neuronal Exosome Markers in Parkinson’s Disease Diagnostics
Anal. Chem., 2020, 92, 13647-13651;
Online Paper
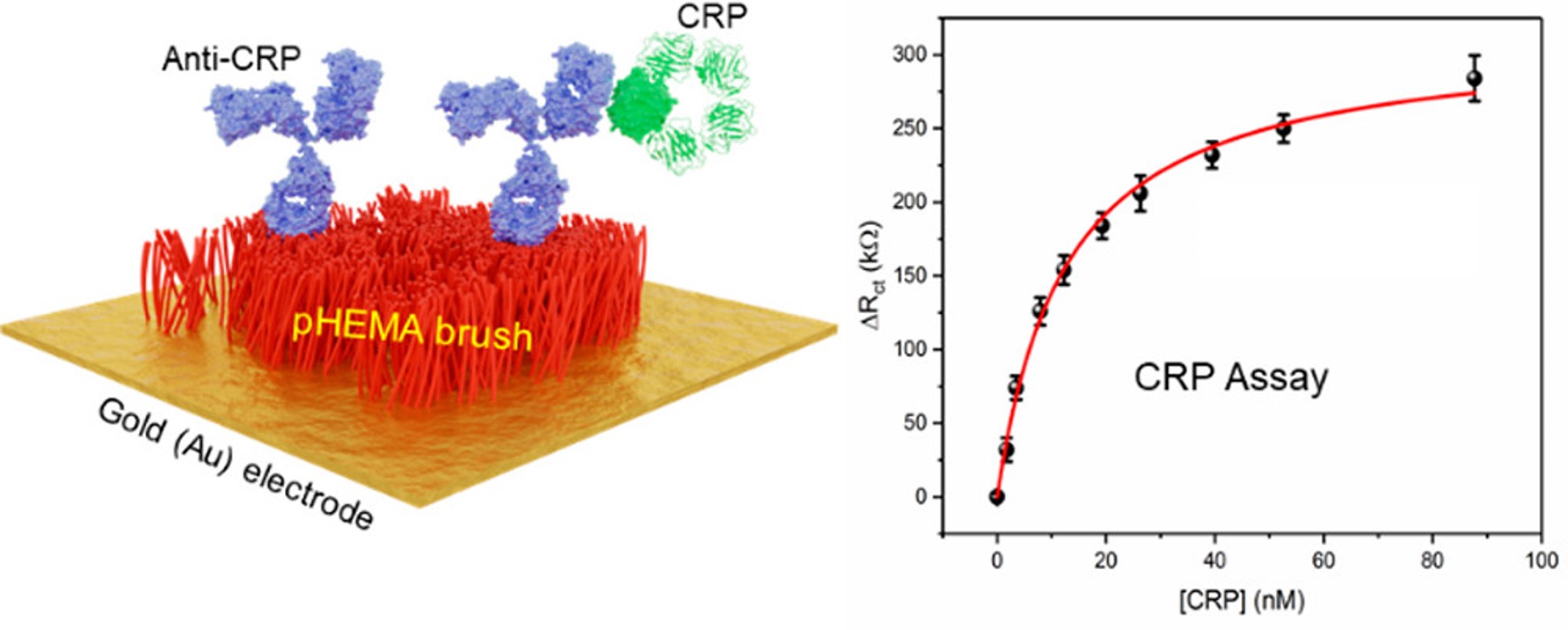
Ultrasensitive Impedimetric Immunosensor for the Detection of C-Reactive Protein in Blood at Surface-Initiated-Reversible Addition–Fragmentation Chain Transfer Generated Poly(2-hydroxyethyl methacrylate) Brushes
Anal. Chem. 2020, 92, 4707-4710;
Online Paper
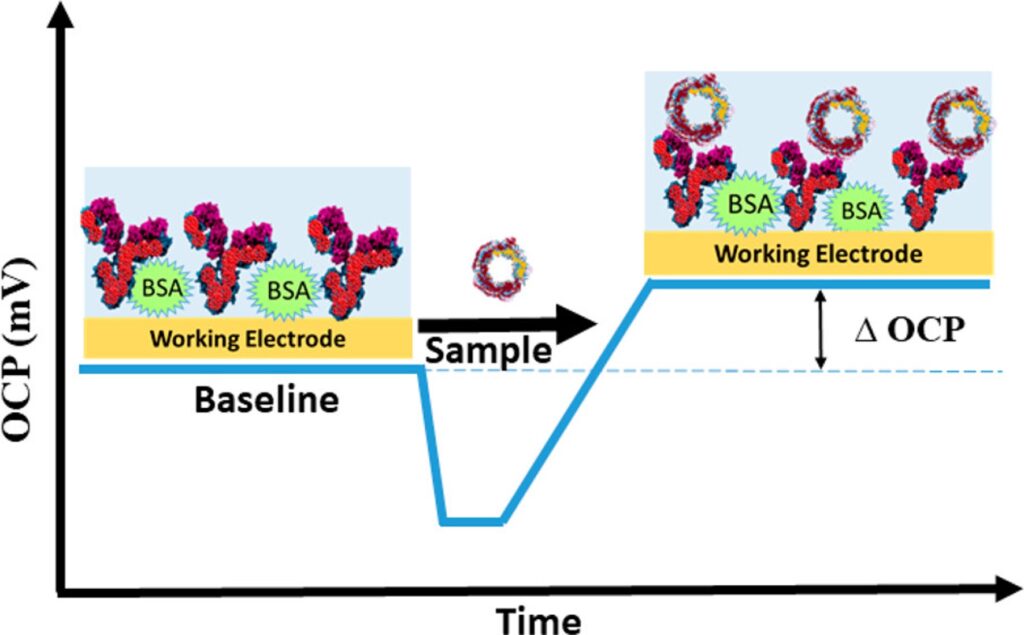
Open Circuit Potential as a Tool for the Assessment of Binding Kinetics and Reagentless Protein Quantitation
Anal. Chem., 2021, 93, 14748-14754;
Online Paper
Graphene Oxide Interfaces in Serum Based Autoantibody Quantification
Anal. Chem., 2015, 87, 346-350;
Online Paper
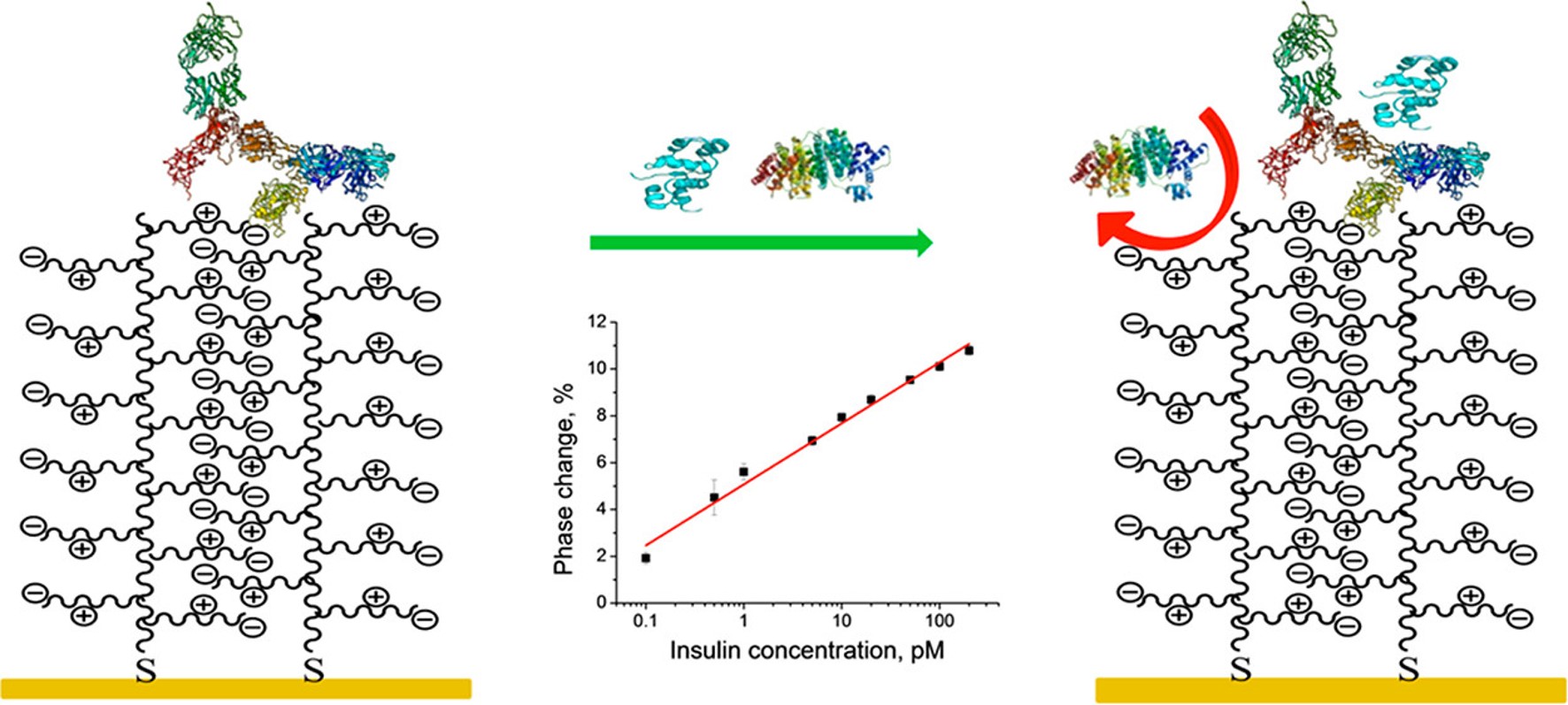
Ultrasensitive Label Free Electrical Detection of Insulin in Neat Blood Serum
Anal. Chem., 2013, 85, 4129-4134;
Online Paper
Electrical biosensors and the label free detection of protein disease biomarkers
Chem Soc Rev., 2013, Vol. 42, 5944-5962
Online Paper
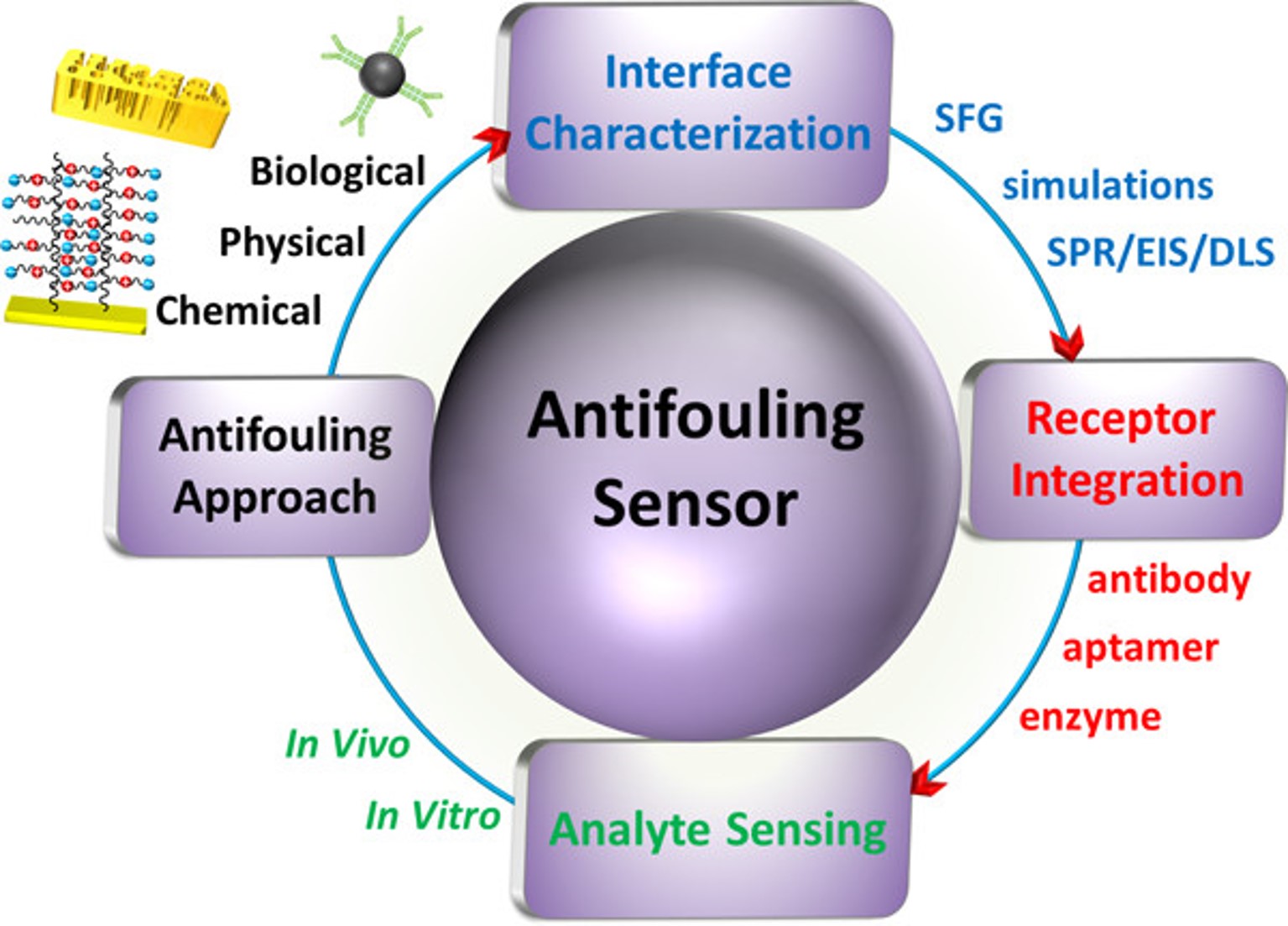
Antifouling Strategies for Selective In Vitro and In Vivo Sensing
Chem. Rev. 2020, 120, 8, 3852–3889
Online Paper
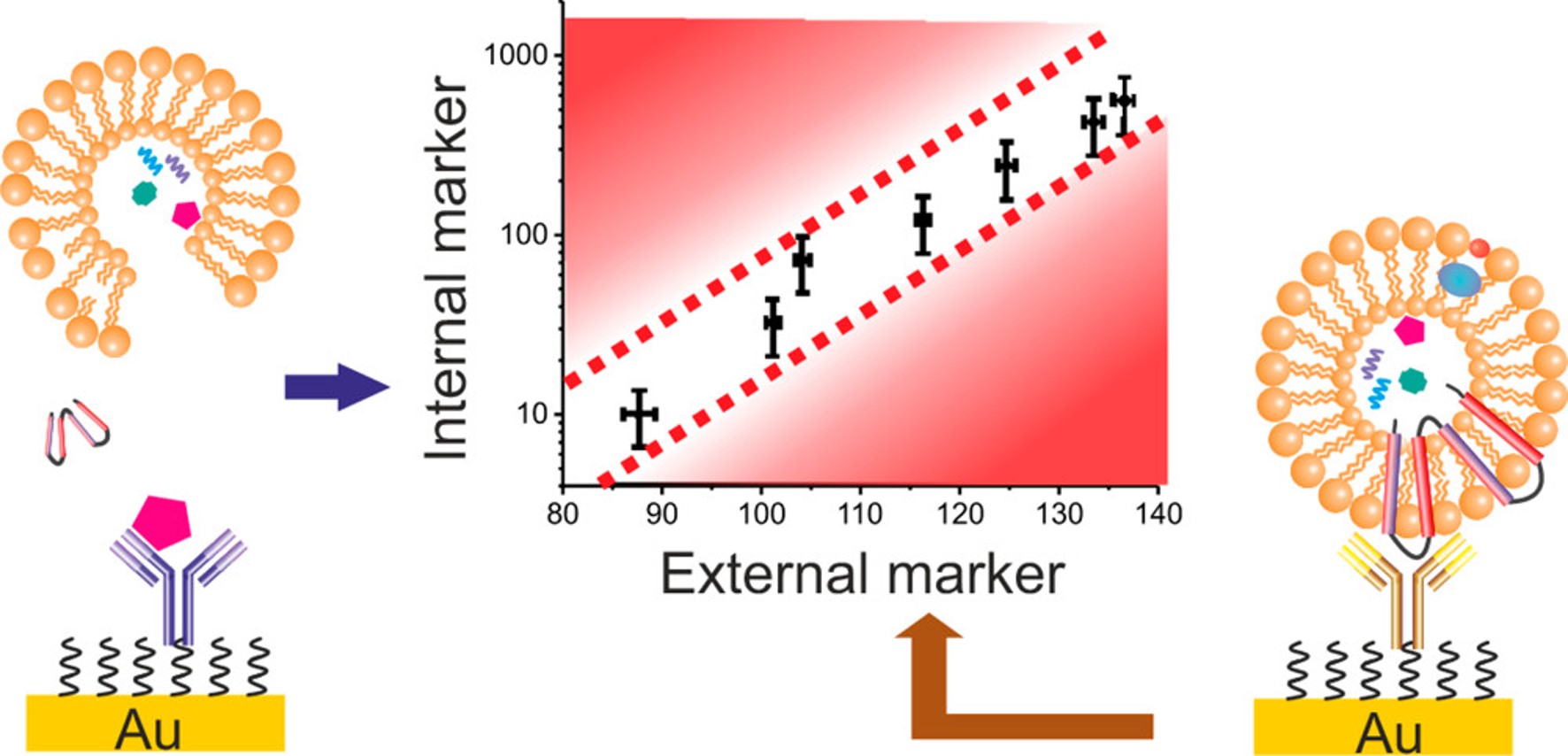
Concentration-Normalized Electroanalytical Assaying of Exosomal Markers
Anal. Chem., 2017, 89, 3184-3190;
Online Paper